If you experiencesudden hearing loss or Meniere’s disease your ENT doctor may want to treat the problem with a locally-applied steroid.
Commonly, he will use: Dexamethasone, Methylprednisolone or Triamcinolone
– Or that is what he tells you.
In reality, the actual forms of drug commonly used for intratympanic injections are:-
Dexamethasone-phosphate
Methylprednisolone-hemisuccinate or succinate
Triamcinolone-acetonide.
In some cases, these alternate forms of the drug have hydrophilic (water-loving), “polar” groups bonded to them, such as the phosphate or succinate groups. This makes the molecule more water-soluble, so the injection can include drug at a higher concentration. In other cases, the drug is modified to add lipophilic groups, such as the acetonide bonded to triamcinolone, which can make it more effective by allowing it to pass though biological membranes more easily.
However, in each of these cases, the drug given is a completely different substance to the “base molecule” and has different molecular properties, as shown in Figure 1. The different forms of the same substance molecules have completely different pharmacokinetic properties in the ear. The two forms of the drug will distribute in the ear in a different way and their effects on the ear will be completely different. The two forms of the drug are not "equivalent". They are completely different drugs, even though they may have the same name.
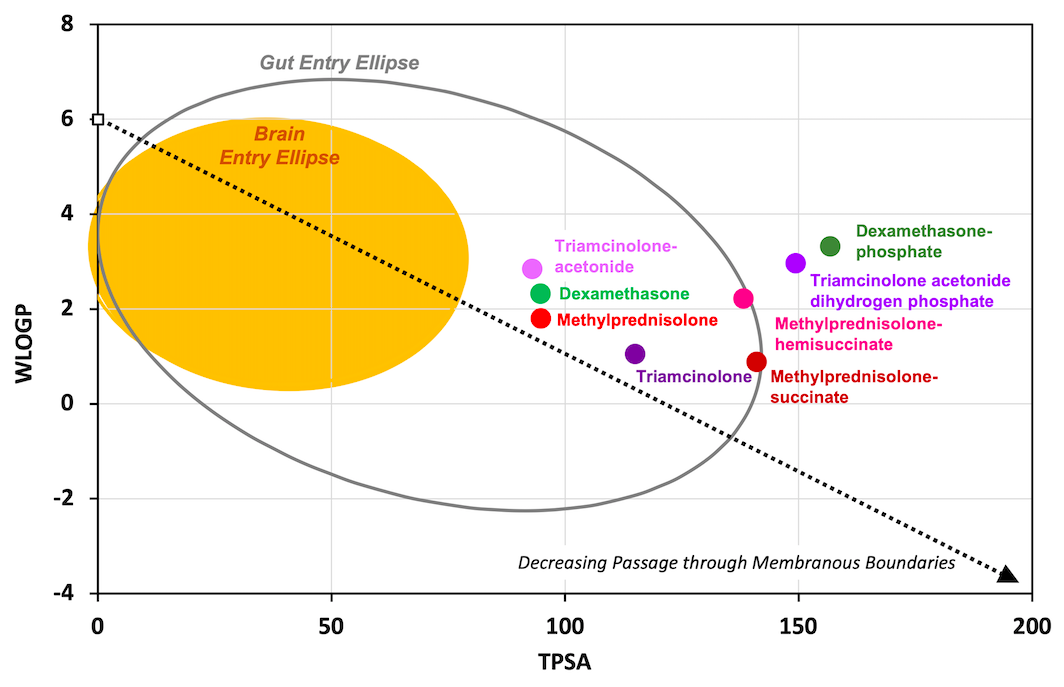
Figure 1: "Egg Plot" showing the physical properties (WLOGP and TPSA) for different steroids applied to the ear. These properties govern how easily drugs ass through biological membranes and profoundly affect how the substances enter and distribute in the ear. Dexamethasone-phosphate is a different molecule and has completely different properties to dexamethasone. Calling dexamethasone-phosphate "dexamethasone" is grossly inaccurate.
The situation is sometimes made worse by poor labeling of the drug vials. On some, the word "Dexamethasone" is given in large, bold font while the "phosphate" is given in smaller font. Many physicians think they are administering dexamethasone when in reality they are not. During presentations I have given on this topic some have been extremely vehement that they are delivering dexamethasone, only to accept defeat when thy have called their nurses or checked with their pharmacy to check the product label carefully.
The problem is that the use of “loose terminology” has carried over to scientific publications. When a paper states that "dexamethasone" was given, this is only correct in very few cases. Far more commonly dexamethasone-phosphate was in fact given. This is glaringly apparent from the method detail when the applied concentration (e.g. 4 mg/mL, 10 mg/mL or 24 mg/mL) is well above the solubility limit for dexamethasone (89 ug/mL). Only dexamethasone-phosphate is soluble at these concentrations. The reader shouldn't have to be uncertain about which drug was given. It is irresponsible to use the incorrect name. The same is true for other steroids. When a paper says triamcinolone was given, in most cases it wasn't. Rather, triamcinolone-acetonide was actually given which has completely different effects compared to triamcinolone. When other groups try to replicate the study and deliver the base form of triamcinolone to the ear (because that was what was stated) they will get completely different results. This will result in total confusion because readers cannot distinguish what was actually given in each study. At the moment, the literature on steroid delivery to the inner ear is an absolute mess, because authors have not defined their drug forms accurately. You are left guessing what drug was actually used.
All of these drugs are in formulations intended for intravenous use. When given intravenously, the exact form the drug has much less significance as they are metabolized to the base form by the liver. But in the ear, it's a totally different story. The specific form affects the rate of elimination from the middle ear, the rate of entry into perilymph through the round window membrane, and the rate of drug loss from perilymph to blood. Each of these properties profoundly affect the amount and distribution of drug in the ear, causing different effects In the ear it is critical to know which form of drug is actually given.
The figure below shows the calculated distribution of triamcinolone-acetonide (left) and triamcinolone (right) along the human cochlea over a 24 hour period. The pharmacokinetic properties of the two drugs in perilymph are completely different, as are the distribution characteristics. They are completely different drugs with different properties. Because of this, it is important to get the name right, otherwise major mistakes will be made.

Conclusion:
Both to patients and in scientific literature - we need to state the exact form of drug being delivered to the ear.
Dexamethasone-phosphate is NOT Dexamethasone
Triamcinolone-acetonide is NOT Triamcinolone
Methylprednisolone-hemisuccinate is NOT Methylprednisolone
This is presented in more detail in our publication:-
Salt AN, Plontke SK. Steroid Nomenclature in Inner Ear Therapy. Otology & Neurotology 2020 41(6):722-726.